Rebecca Yao commenced her PhD in 2015 with the aim of her project to determine new methods to purify olfactory cells useful for transplantation to repair the injured spinal cord. While olfactory cells have been shown to partially regenerate the injured spinal cord, improvements in the therapy outcomes can be achieved by if higher quality cell preparations are obtained.
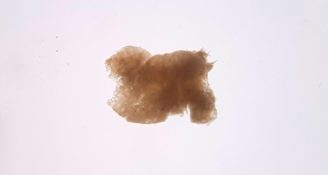
The difficulty is that the biopsy of olfactory tissue that is taken from the patient has a wide range of different cells that need to be first separated and then encouraged to increase in number to product sufficient cells needed for transplantation.
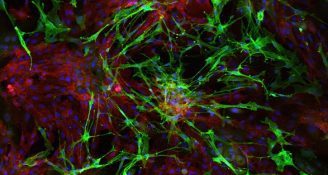
Rebecca’s PhD project directly addressed this issue, by testing new methods for purifying the cells to determine which method generates the highest purity of cells. Thanks to the funding by the Count Charitable Foundation, Rebecca was been able to test a range of different commercial options for purifying the cells, and compared them to existing methods that are currently used by scientists around the world.
Her results were both surprising and encouraging. Rebecca discovered that the current methods do not produce sufficient purity because the identity of the cells has been inaccurate. To overcome this, she has created a panel of markers to better identify the cells that are needed, which is the important first step in the process. She then compared and tested many different methods for growing cells in the lab and produced an optimal protocol which consistently gives high purity of the target cells.
Due to the large Spinal Injury Project team at Griffith University, other members of the team are now using the improved purification method to produce the high quality cells needed for regenerating the spinal cord.
This project was kindly sponsored by Count Charitable Foundation.
Publication
Yao R, Murtaza M, Tello Velasquez J, Todorovic M, Rayfield A, Ekberg J, Barton J, St John J. 2018. Olfactory ensheathing cells for spinal cord injury: sniffing out the issues. Cell Transplantation, January 1-11, DOI: 10.1177/0963689718779353.